DUVYZAT protects from Duchenne progression
The clinical trial measured whether DUVYZAT + steroids more effectively preserved muscle strength and function than steroids alone (placebo). See more about the trial >
Completed 4-stair climb 1.78 seconds faster
than participants taking steroids alone, demonstrating significant protection.*
*This was measured by the average time it took participants in both groups to climb 4 stairs at the end of 18 months.

Additional measurements of protection
After 18 months, participants taking DUVYZAT + steroids:
-
Maintained more ability to bend their elbow or straighten their knee, a measure of muscle strength
-
Maintained motor function abilities longer compared with those who took placebo, based on North Star Ambulatory Assessment (NSAA) scores
- The NSAA is a 17-item test that looks at everyday movements, such as ability to stand up from the floor, walk, climb stairs, balance, and jump
- Those who took DUVYZAT + steroids saw an average of 1.91 fewer points of decline on the NSAA than those who took only steroids
-
DUVYZAT also reduced how much new fat was in the muscle, a measurement of muscle loss
- Those who took DUVYZAT + steroids had 30% less new fat build-up in their muscles than those who were on steroids alone
DUVYZAT reduced how much new fat was in the muscle
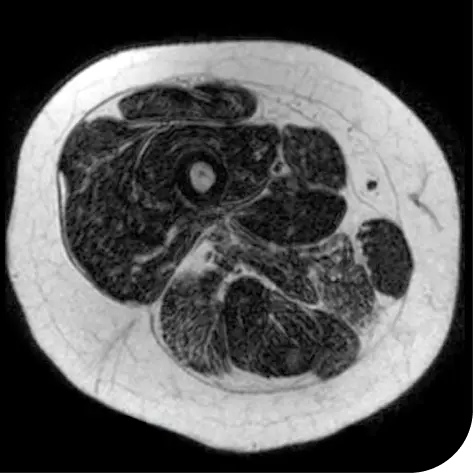
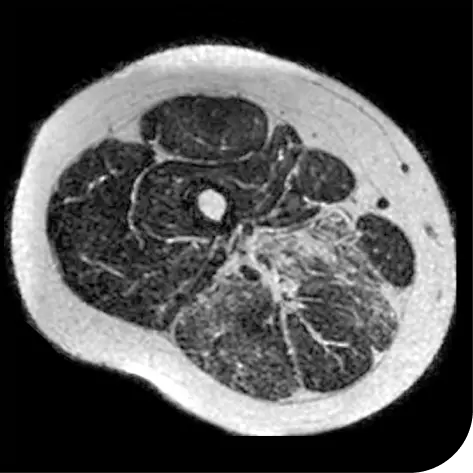
In another follow-up analysis of the clinical trial (meaning it was not one of the main measures planned when the study began), researchers looked at individual people to see how much new fat was in the muscle. The images below represent results from 2 individual people from the study after 18 months. These results may not be the same for everyone.
How to read these images:
- Magnetic resonance spectroscopy (MRS) images can show the difference between muscle and fat in the body
- By taking images of the same patients 18 months apart, researchers can track the amount of new fat that is building up in the muscles
- The darker areas of the images represent muscle tissue and the white areas represent fat
| At the beginning of the clinical trial |
After 18 months | |
 |
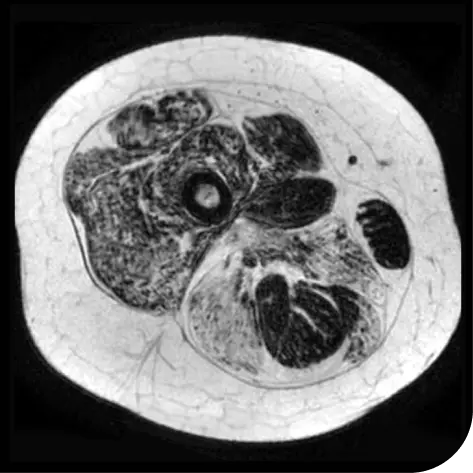 |
Participant taking steroids alone |
 |
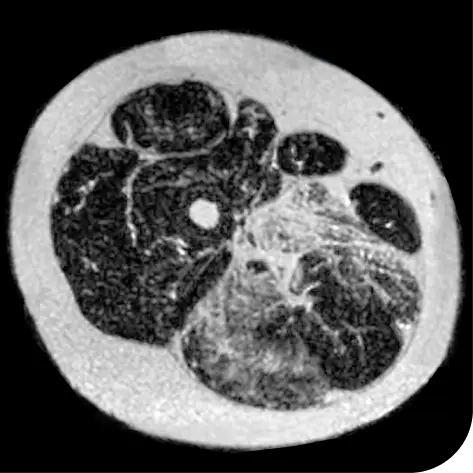 |
Participant taking DUVYZAT + steroids |

How DUVYZAT was studied
The clinical trial was 18 months long
- During the trial, the DUVYZAT dose was based on weight and was reduced if certain side effects occurred
- The effectiveness of DUVYZAT was studied in 120 participants
- The safety of DUVYZAT was studied in 179 participants
SEE WHAT THE RESEARCHERS LOOKED AT

4-stair climb
How fast participants could climb 4 stairs before and after 18 months of using DUVYZAT and steroids compared with steroids alone

NSAA
Participants’ mobility and muscle function across multiple muscle groups as measured by the NSAA, a test that measures how well people with Duchenne can move and do daily activities

Muscle strength
A muscle strength test that included how well participants could bend their elbow and straighten their knee measured with a small handheld device called a myometer

Fat tissue
Measurement of how much fat was in the muscle of the upper leg to understand muscle loss. This was assessed by MRS, an imaging test
